Jim Rosinski
Jim Rosinski da Fundação CHDI falou sobre a estratégia -omics na Fundação CHDI para identificar biomarcadores precoces da progressão da doença, com foco hoje na análise precoce do LCR e amostras de plasma derivadas da DH estudo de clareza usando o somalógica plataforma. O objetivo da estratégia é aplicar múltiplas plataformas para identificar/alinhar biomarcadores de progressão da doença que possam ser transformados em ensaios quantitativos validados para uso em programas clínicos. O Plataforma Enroll-HD, dada a sua dimensão e natureza longitudinal, constitui a base de toda a estratégia -omics. Jim explicou algumas das plataformas tecnológicas atuais que estão sendo implantadas para análise de sangue e LCR, incluindo detecção baseada em especificação de massa, análise de pequenos RNA, plataformas de proteômica Somalogic e epigenética, metilação, análise de células sanguíneas, entre outras.
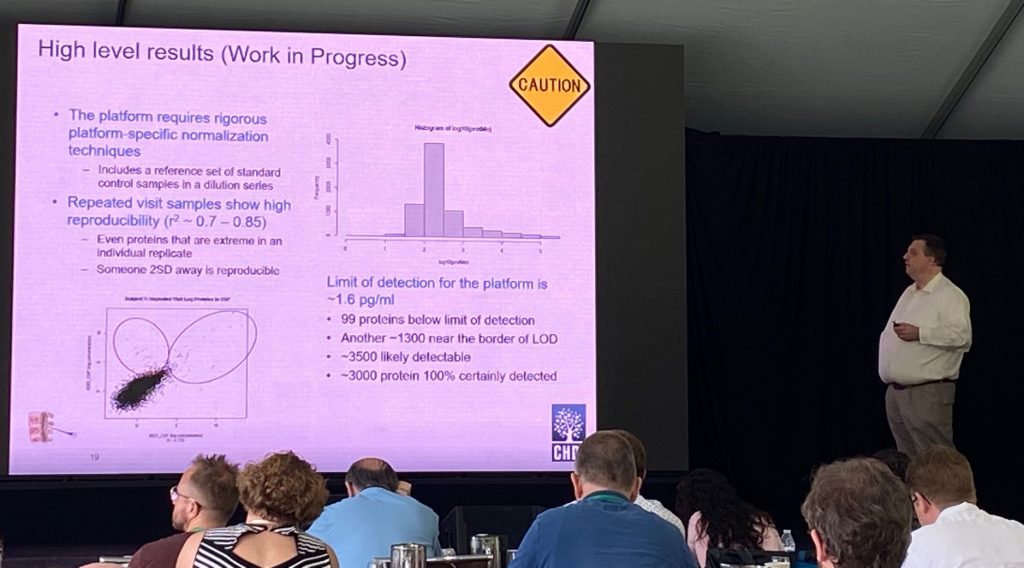
Uma coleção de 579 amostras de CSF e soro foram analisadas do estudo HD Clarity (420 participantes, 150 amostras repetidas para replicação) usando a plataforma Somalogic baseada em aptâmero (DNA) (7.629 proteínas sendo medidas, embora HTT não esteja em sua plataforma). Jim descreveu a análise inicial preliminar dessas amostras usando essa tecnologia. A análise das “visitas repetidas” (1 mês de intervalo) mostrou uma correlação muito boa (r=0,7-0,85) detectando cerca de 3500 proteínas no LCR. A análise foi feita por Doug Langbehn, da Universidade de Iowa.
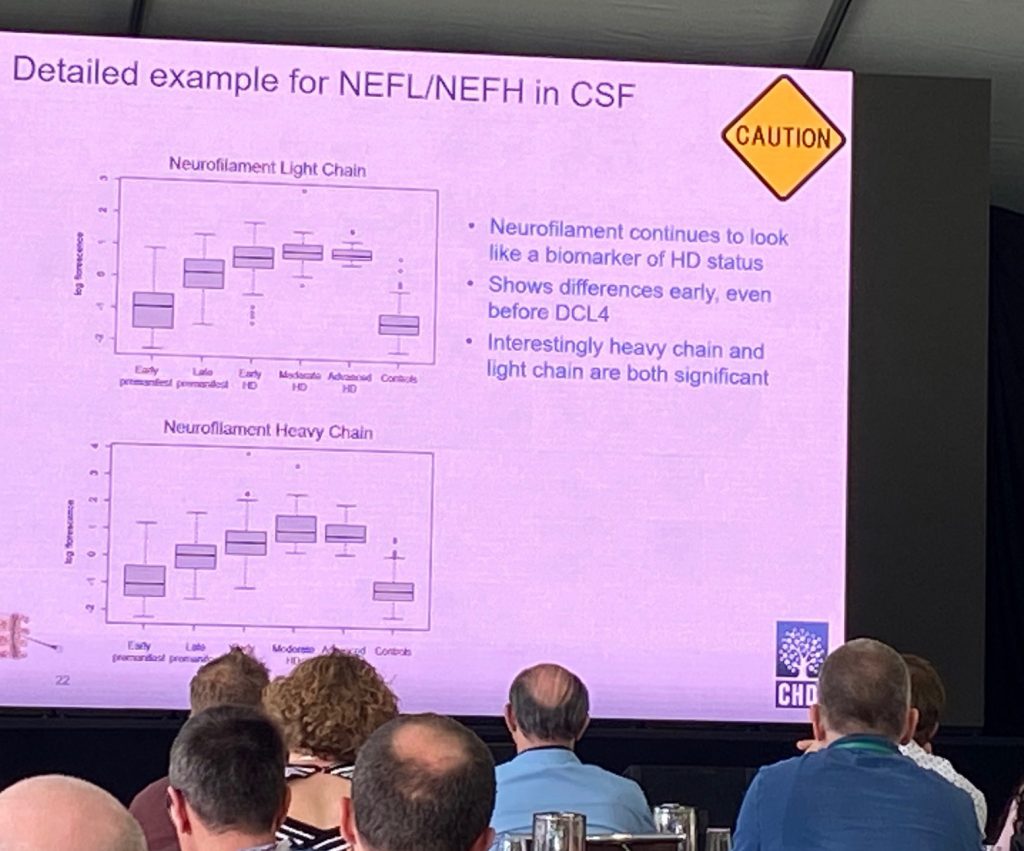
As análises iniciais dos dados mostraram que há um bom número de proteínas (97) sendo diferentes no LCR e no soro com FDR<0,05, principalmente as proteínas de neurofilamentos (NF) NF-leve e NF-pesado. Ambos mostram diferenças significativas desde o início do pré-manifesto. De fato, essas duas proteínas NEFL/NEFH podem prever o estado genético dos participantes e estão altamente correlacionadas com os escores de CAP e cUHDRS. Uma proteína adicional de interesse do CNP (2', 3'-nucleotídeo cíclico 3' fosfodiesterase), uma enzima expressa principalmente em oligodendrócitos, e pode ser um bom marcador de desmielinização ou degeneração axonal. A análise de enriquecimento do proteoma CSF alterado nestas amostras mostra que a assinatura é uma assinatura HD, comumente compartilhada com modelos de camundongos HD, incluindo alterações proteômicas/genéticas Q175 e R6.2. As alterações séricas são menos impressionantes, mas ainda há uma assinatura que pode prever o controle de cUHDRS usando apenas 5 proteínas.
Aline Delva
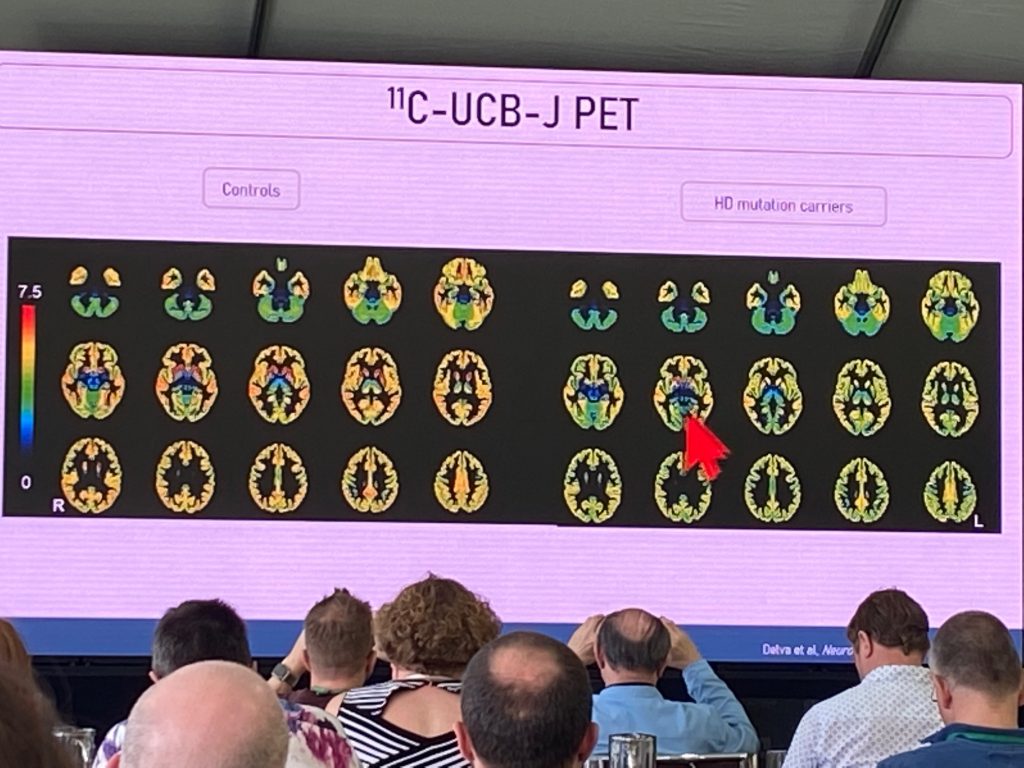
Aline Delva da Universidade de Leuven, falou sobre a primeira avaliação de um traçador de imagem HTT mutante [C11] -CHDI'180R, desenvolvido pela CHDI Foundation, em pacientes em HD. Além disso, ela descreveu o trabalho realizado para imagens de mudanças terminais pré-sinápticas usando o traçador Sv2a PET [C11] SCU-J, em indivíduos com DH precoce. Ela começou primeiro com uma descrição da perda precoce de sinapses em tecidos pós-morte de HD e em modelos de HD. O uso desse novo marcador pode ser indicativo de alterações degenerativas no cérebro de pessoas em vários estágios da doença e também pode atuar como um marcador de estratégias regenerativas ou neuroprotetoras sinápticas. Aline também incluiu [F18]FDG-PET em um estudo inicial para comparar mudanças na densidade sináptica com atividade funcional e com ressonância magnética estrutural. Num estudo publicado recentemente, foram incluídos um total de n=18 portadores de DH (n=7 pré-manifestos en=11 manifestos) e n=15 controles.
As alterações do FDG-PET se correlacionam bem com as alterações do UCB-J em todo o cérebro. As assinaturas de hipometabolismo replicam alterações anteriores, embora o sinal UCB-J sugira alterações generalizadas na densidade sináptica no córtex frontal-temporal-parietal, gânglios da base e cerebelo, evidentes já no estágio pré-manifesto. O UCB-J foi o marcador mais sensível nos estágios pré-manifestos e se correlaciona extremamente bem com o escore motor UHDRS e a tarefa SDMT. A pontuação motora foi melhor correlacionada com as alterações de ligação do UCB-J no putâmen. Este é um marcador muito significativo e sugere fortemente que a DH precoce envolve alterações pré-sinápticas significativas, começando no estriado (provavelmente fibras aferentes provenientes de regiões corticais ou talâmicas) e depois progredindo para regiões extra-estriatais.
Em seguida, Aline descreve o primeiro trabalho em humanos realizado com os novos rastreadores de imagem PET específicos para mHTT, em colaboração com a CHDI Foundation. Ela resumiu o trabalho pré-clínico anterior conduzido pelo CHDI e os resultados dos estudos de dosimetria humana, que mostraram que [C11] -CHDI'180R pode avançar para estudos clínicos de DH devido à sua segurança, cinética cerebral e perfis metabólicos.
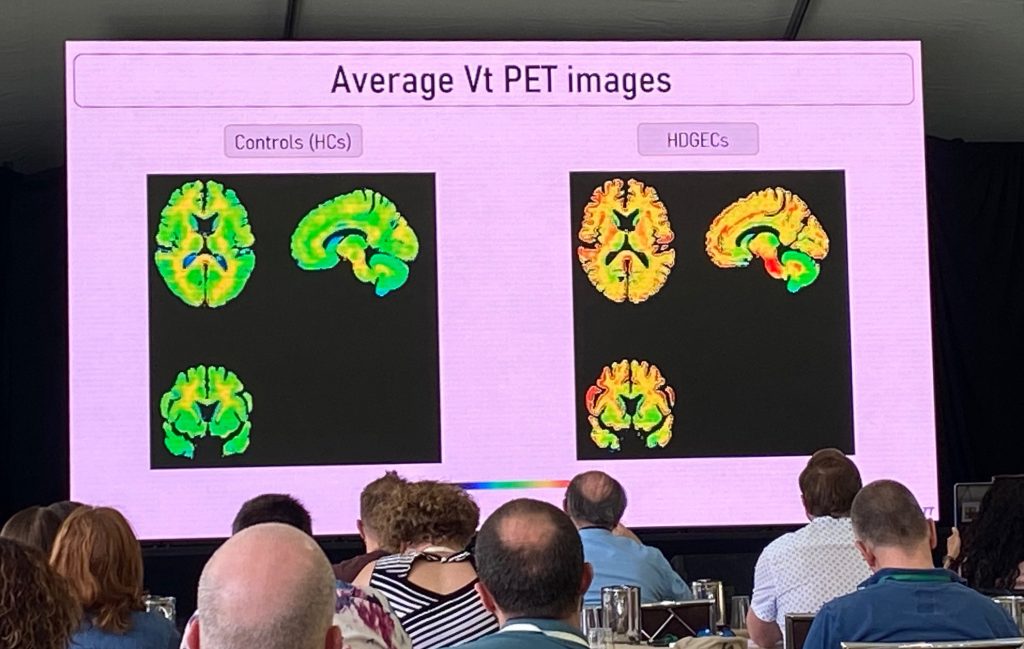
O estudo ImageHTT é o primeiro a avaliar diferenças em [C11] -CHDI'180R ligando-se ao cérebro HD versus cérebros de controle no primeiro estudo desse tipo. Os resultados da fase 1 deste estudo foram discutidos, onde alguns jovens controles versus pacientes em HD em estágio 1-2 (n=6 por grupo) foram avaliados. Estudos adicionais envolvendo mais indivíduos em HD e mais pareados por idade serão incluídos, conforme descrito abaixo.

O volume médio de distribuição (Vt) é maior em HD versus controles, embora haja uma variabilidade considerável entre os vários sinais e, portanto, as diferenças globais entre os grupos não foram significativas. Usando uma técnica diferente (DVR médio, proporções de volume de distribuição), foi obtida significância estatística para algumas regiões corticais, embora os efeitos sejam pequenos. Mais trabalho é necessário (confiabilidade teste-reteste, mais sujeitos e diferentes estágios de DH), mas as descobertas iniciais são animadoras. Existem novos radioligantes adicionais que estão surgindo, como h [C11] -CHDI'009, que tem uma ligação muito maior em estudos pré-clínicos.